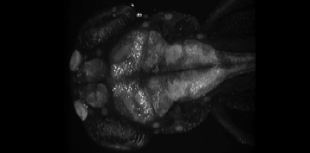
Research Areas
Myopathy
Neuropathy
ADHD
We investigate the genetic causes, mechanisms of disease, and potential therapies for a range of muscle diseases (myopathies), using the advantages of the zebrafish model system for in vivo imaging, genetic manipulation, and screening to accomplish these goals.
The process of muscle development, and the genes involved, are highly conserved between zebrafish and humans, therefore the zebrafish provides and excellent model system in which to study myopathies. We have previously utilised zebrafish to provide insight into muscular dystrophies and congenital myopathies.
We are currently investigating a number of myopathies including nemaline and myofibrillar myopathies as described below.
Myopathy

Nemaline Myopathy
Nemaline myopathy is a congenital myopathy characterised by muscle weakness and the presence of rod-like structures within the muscle termed nemaline bodies. There is great variation in the severity of nemaline myopathies ranging from complete loss of movement at birth to mild muscle weakness in adults. Several mutations have been identified to cause nemaline myopathy with most of the identified cases being dominanlty inheritied. Despite the identification of the genetic cause how the nemaline bodies or mutant proteins result in muscle weakness is not understood. We are creating zebrafish models of these muscle diseases to allow us to investigate how these mutations results in disease and identify potential therapies for these disorders.
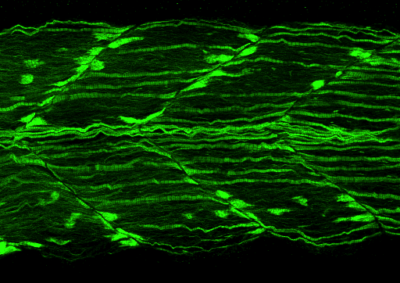
Myofibrillar Myopathy
Myofibrillar myopathies are a group of late onset muscle diseases with progressive muscle weakness. At the cellular level myofibrillar myopathies are characterised by formation of protein aggregates and disintegration of the muscle fibre. So far mutations in seven genes have been shown to result in myofibrillar myopathy but there remain many cases for which the mutation has not been identified. Through the investigation of zebrafish mutant strains with muscle defects we aim to identify further genes involved in these disease. Whilst the genetic causes are known in many cases the mechanism by which mutations in these genes result in disease are poorly understood. We use the advantages of the zebrafish model system, allowing genetic manipulation and in vivo imaging to investigate the onset and progression of disease and determine the processes that are disrupted resulting in muscle weakness. We have used our models to identify that autophagy is disrupted in many forms of myofibrillar myopathy, a finding we have confirmed in patient samples. As a result we condicted driugs screen with autophagy promoting drugs and identified metfomin as a drug that may be